|
Understand why, within a covalently-bonded. Electron geometry: Electron pairs need to be as far away as possible in order to minimize repulsions. This interactive activity from ChemThink explains the valence shell electron pair repulsion (VSEPR) theory. But this model does not say anything regarding the multiple bonds present. 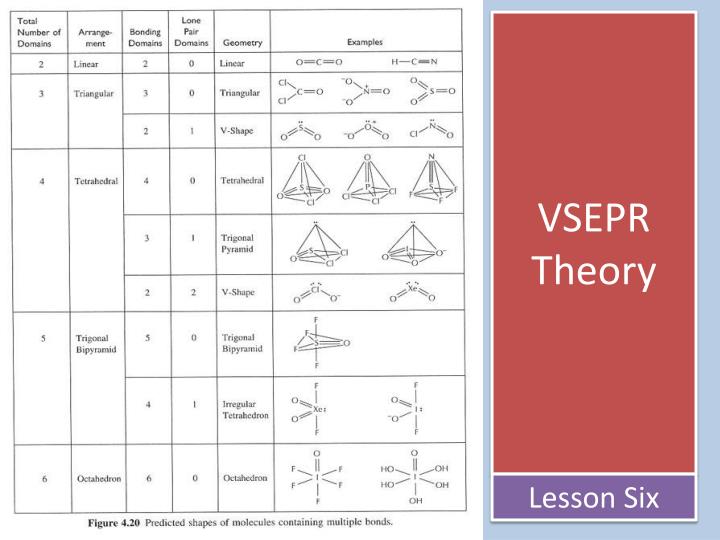
Two components: electron geometry and molecular geometry. VSEPR model helps to understand the different shapes and arrangement of molecules. The valance shell electron pair repulsion theory states. If e- are not shared evenly btn 2 atoms, the atom with a stronger pull on e- (higher EN) becomes slightly –ve and the other atom becomes slightly +ve this is called a dipole and can be drawn in 2 waysĮxamples of Dipoles Gr. Valence-shell electron-pair repulsion theory because electron pairs repel, molecules adjust their shapes so that valence electron pairs are as far apart as possible. Same topic, but a simpler video.This video explains VSEPR Theory & Molecular Shapes.∆EN Covalent 0 – 0.5 Polar Covalent 0.5 – 1.7 Ionic > 1.7 What is the VSEPR theory used to predict answer choices. NOTE: noble gases have no # indicating a value of 0.Check difference in electronegativity btn the 2 atoms The VSEPR predicted shapes of molecules can be found in a systematic way by using the number of electron pairs to determine the shape of the molecules.Ionic: exchange of e- btn 2 atoms Covalent: equal sharing of e- btn 2 atoms Polar Covalent: unequal sharing of e- btn 2 atoms
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
